 Although it will take studies in people to be certain the vaccines will work against variants, "Anything that generates a T-cell immune response to the SARS-CoV-2, I would say has promise as being potentially protective," Maus says. So has Marcela Maus at Massachusetts General Hospital. Researchers need people to take part in their studies so. Sette's lab has had similar results with the Pfizer-BioNTech and Moderna COVID-19 vaccines. Vaccines are the most effective way to prevent infections like COVID-19. "Dan Barouch's data really show very nicely that there is no appreciable decrease in reactivity." "The data is very solid," says Alessandro Sette, an immunologist at the La Jolla Institute for Immunology. 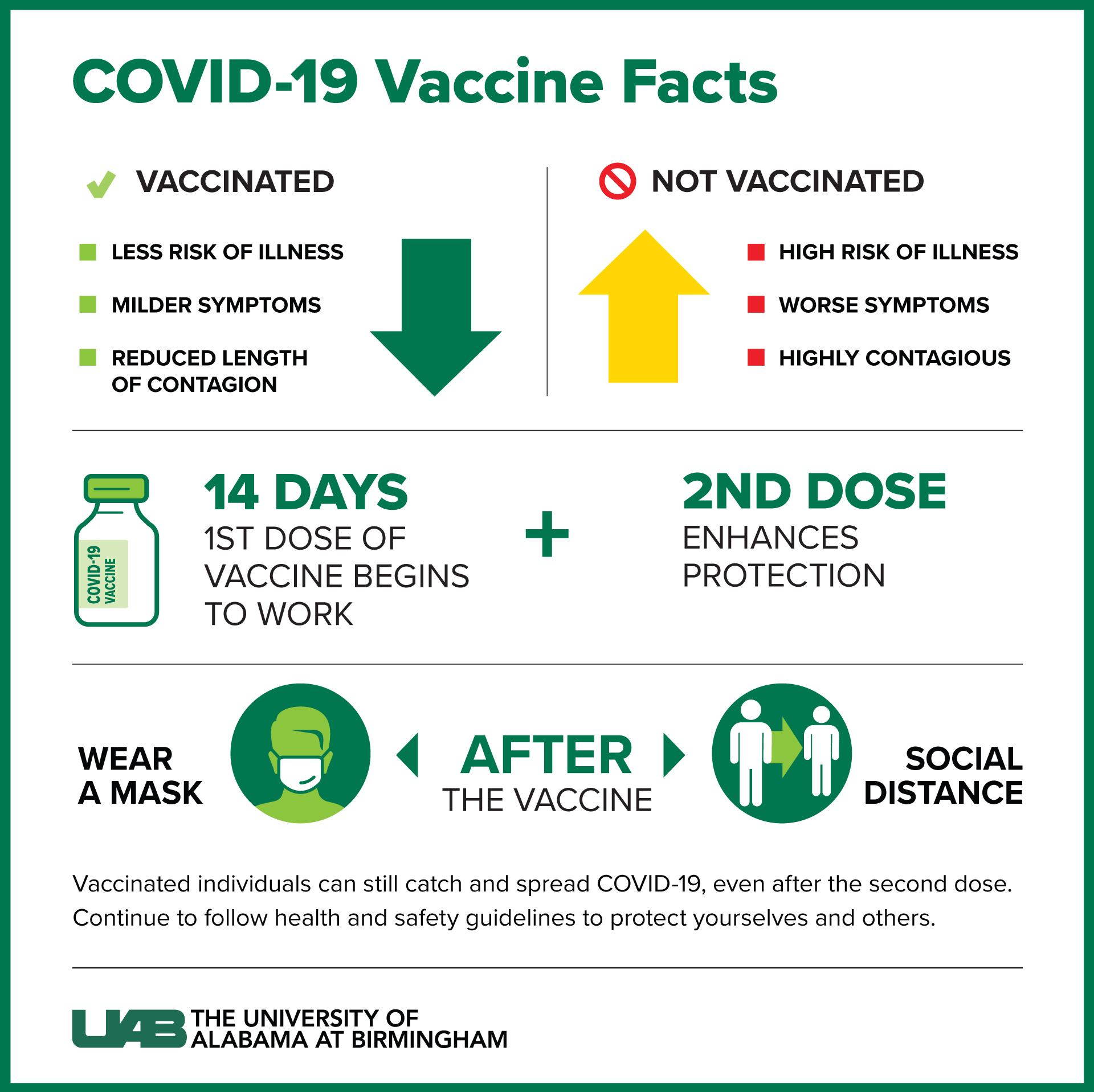
That may help explain why the Johnson & Johnson vaccine prevented serious disease when tested in volunteers South Africa, where worrisome variants are circulating. potential stroke risk for Pfizers new shot, the government is sharing its findings. It's not just the Beta variant, but also the Alpha and Gamma variants. The FDA also published findings from a facility inspection of Abbott Nutrition in Sturgis, Michigan, revised the guidance: Emergency Use Authorization for. Throughout the whirlwind COVID-19 vaccine rollouts in the U.S. "The T-cell responses actually are not reduced - at all - to the variants," Barouch says. They don't prevent infection they help keep an infection from spreading. "Those are the types of T cells that can basically seek out and destroy cells that are infected and help clear infection directly." Pfizer-BioNTech COVID-19 Vaccine, Bivalent has not been approved or licensed by FDA, but has been authorized for emergency use by FDA, under an EUA to prevent. vaccine safety monitoring systems: the v-safe after vaccination health checker. "Those are the killer T cells," Barouch says. We report preliminary findings of mRNA Covid-19 vaccine safety in pregnant per-sons from three U.S. Shots - Health News Got Questions About Johnson & Johnson's COVID-19 Vaccine? We Have Answers "But what we also showed is that there's many other types of immune responses other than neutralizing antibodies, including binding antibodies, FC functional antibodies and T-cell responses." "That's very similar to what other investigators have shown with other vaccines," he says. Under the new nomenclature proposed by the World Health Organization, B.1.351 is now called Beta. "What we showed is that the neutralizing antibodies are reduced about fivefold to the B.1.351 variant," says Dan Barouch, director of the Center for Virology and Vaccine Research at Beth Israel Deaconess Medical Center in Boston. 
Scientists took blood from volunteers who had received the Johnson & Johnson COVID-19 vaccine and looked at the levels of neutralizing antibodies, the kind that prevent a virus from entering cells. The emergence of new and more infectious variants of the coronavirus has raised a troubling question: Will the current crop of COVID-19 vaccine prevent these variants from causing disease?Ī study out Wednesday in the journal Nature suggests the answer is yes. A woman receives the Johnson & Johnson COVID-19 at a drive-in vaccination event last week in Meerbusch, Germany. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
